Going from Paper to Digital without any chaos
Unified Operations, Uncompromised Integrity without Silos

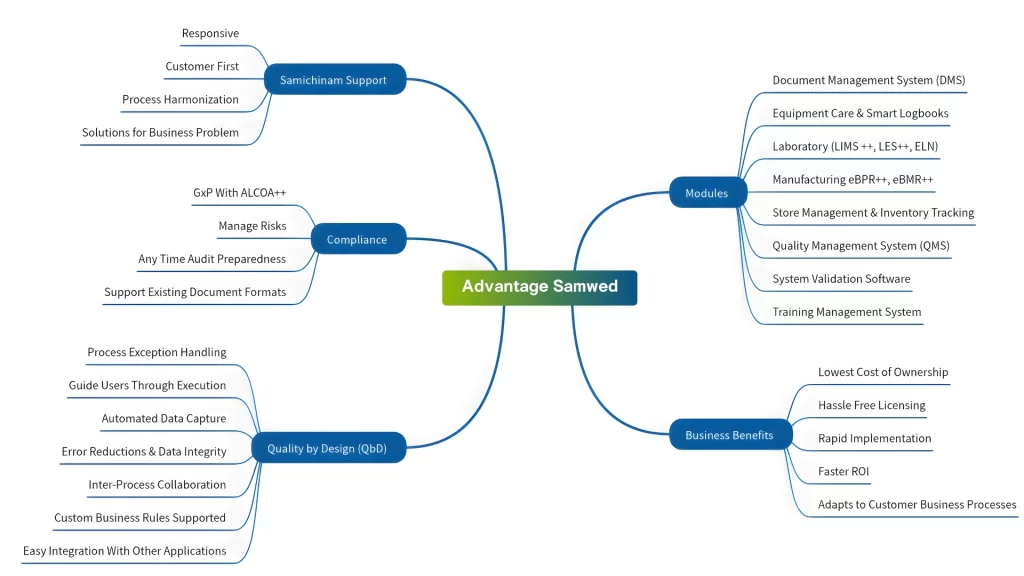
Accelerate your journey to Pharma 4.0 with Samwed Suite
Samwed is an advanced AI enabled process engine designed to unify your pharmaceutical plant’s operations—from warehousing and manufacturing to quality assurance (QA). Built for speed and compliance, Samwed bridges the gap between paper-based systems and total digital automation. With agile deployment support from our Customer Success team, we ensure data integrity, seamless process harmonization, and rapid user adoption across your entire organization.
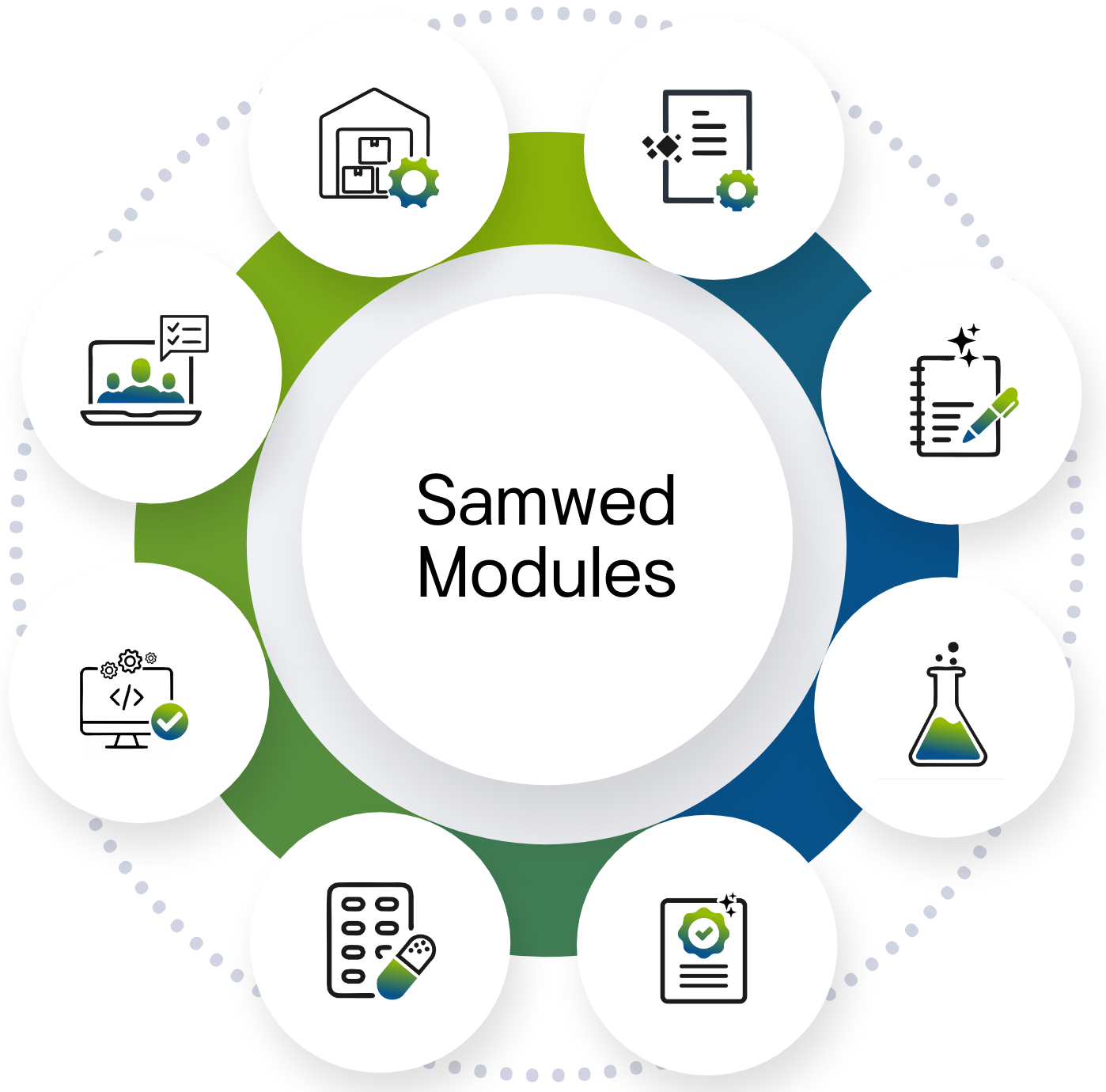
Samwed Modules
Your Roadmap to Risk Mitigation
One Platform, Infinite Choice. Your Transformation, Your Timeline
Dive in with a single module whether LIMS today or QMS tomorrow. Let us expand your Samwed ecosystem only when you are ready. You don’t have to digitize everything at once. We handle the configuration to ensure a smooth, step-by-step transition with a tailored Digital Transformation Roadmap aligned with your business goals.
Be Audit-Ready by Design. Eliminate the risk of FDA Form 483s. Samwed is built on Quality by Design (QbD) principles, embedding regulatory compliance directly into your workflows so your processes are always audit-ready. Automated Documentation & SOPs -Minimize manual paperwork instantly. Save documentation effort and let Samwed create your SOPs, reports etc automatically from the configured process.
Master Quality by Design (QbD) with Samwed
Uncompromised Data Integrity
Automated Data Acquisition
Guided Digital SOPs
Automated Exception Handling
Seamless ERP Integration
Frequently Asked Questions (FAQ's)
The organization that wants to grow globally, needs to be scalable and comply to all the regulations of the specific market where they want to grow. With increasing conditions from regulators across the world, paper or silos fail scalability, data integrity and adherence to ALCOA++ principles.
The organization needs a digital platform which will create a unified digital twin of your process.
Smart digital systems eliminate manual errors, improve data integrity, and provide real-time visibility. With Samwed Suite, you gain configurable workflows, complete audit trails, and ALCOA++ compliance that paper systems cannot offer, while reducing storage costs and retrieval delays by up to 90%.
Samwed is built specifically for GxP environments with compliance to 21 CFR Part 11, EU Annex 11 & adherence to ALCOA++ principles, built in. Our systems include electronic signatures, complete audit trails, automated version control, and validation ready documentation to ensure seamless regulatory acceptance.
We digitize the latest versions of all required documents, through structured scanning, indexing, and metadata tagging, ensuring uninterrupted business process continuity and subsequently enable fully integrated digital workflows within the Samwed Document Management System.
Now your records are searchable, retrievable within a click and no delays.
Typical implementation ranges from 8-16 weeks depending on scope. Our phased approach includes:
current data and process assessment (2 weeks), configuration (4-8 weeks), validation (2-4 weeks), and go-live with parallel run. We provide dedicated project management throughout.
Samwed offers a modular implementation approach.
While we recommend an ideal sequence as DMS → Equipment Care → Laboratory & Manufacturing(eBMR/BPR) → System Validation → Training → QMS to ensure optimal efficiency, we remain flexible and can adapt the implementation order based on your organization’s priorities and requirements.
All modules integrate seamlessly within Samwed when added and with 3rd party systems as required.
Yes, Samwed functions as a business process engine that adapts to your existing workflows, creating a true digital twin of your operations. With deep domain expertise, we not only mirror your current processes to ensure a smooth transition with minimal disruption but also recommend practical harmonizations that enhance efficiency and unlock the full benefits of digitization.
Yes, Samwed offers APIs based integrations with 3rd party software like ERP, LIMS, and MES systems etc
The learning curve for Samwed is minimal for a simple reason that Samwed is Business Process Engine that adapts to your process and creates its digital twin. In addition to this, we provide training programs for every implementation including:
- Method adaption programs with key members to make a smooth transition from paper to digital.
- Instructor-led sessions for administrators and IT team
- “Digital champions”(Train the trainers) program for internal trainers
- On-screen guidance and context-sensitive self-help within the platform
- Telephonic and on-site support as required, post implementation.
Through features like:
- Auto-calculations by Samwed in batch records, TDS etc. where possible.
- Auto fetched values from master data available e.g. product meta data directly fetched from product master.
- Configurable validations for user entries where required.
- Guided selections from lists eliminating typo mistakes in free text.
- Display of expected value/ range to guide users through process
- Integration and duplex communication with equipment/instruments to eliminate manual transcription
- Data entry mandates wherever required.
- AI capabilities to translate the description in User’s native language to eliminate possibility of gaps in understanding the information.
- QR code based scanning to replace data entry of areas / equipment ID etc.
Smart platforms like Samwed are implemented and configured with foundation of Quality by Design and built in compliance to 21 CFR part 11 and ALCOA++ principles. These platforms make sure that the organization is always audit ready.
Digital systems simplify audits with:
- Google like search and filters for quick retrieval of any data and/or document.
- Complete electronic audit trails for necessary user and system actions.
- Real-time reporting dashboards
- Remote audit capabilities
- Automated compliance status reports
- Single Screen audit.